Andros
Kraken
- Joined
- Jan 14, 2020
- Posts
- 3,701
- Reputation
- 6,125
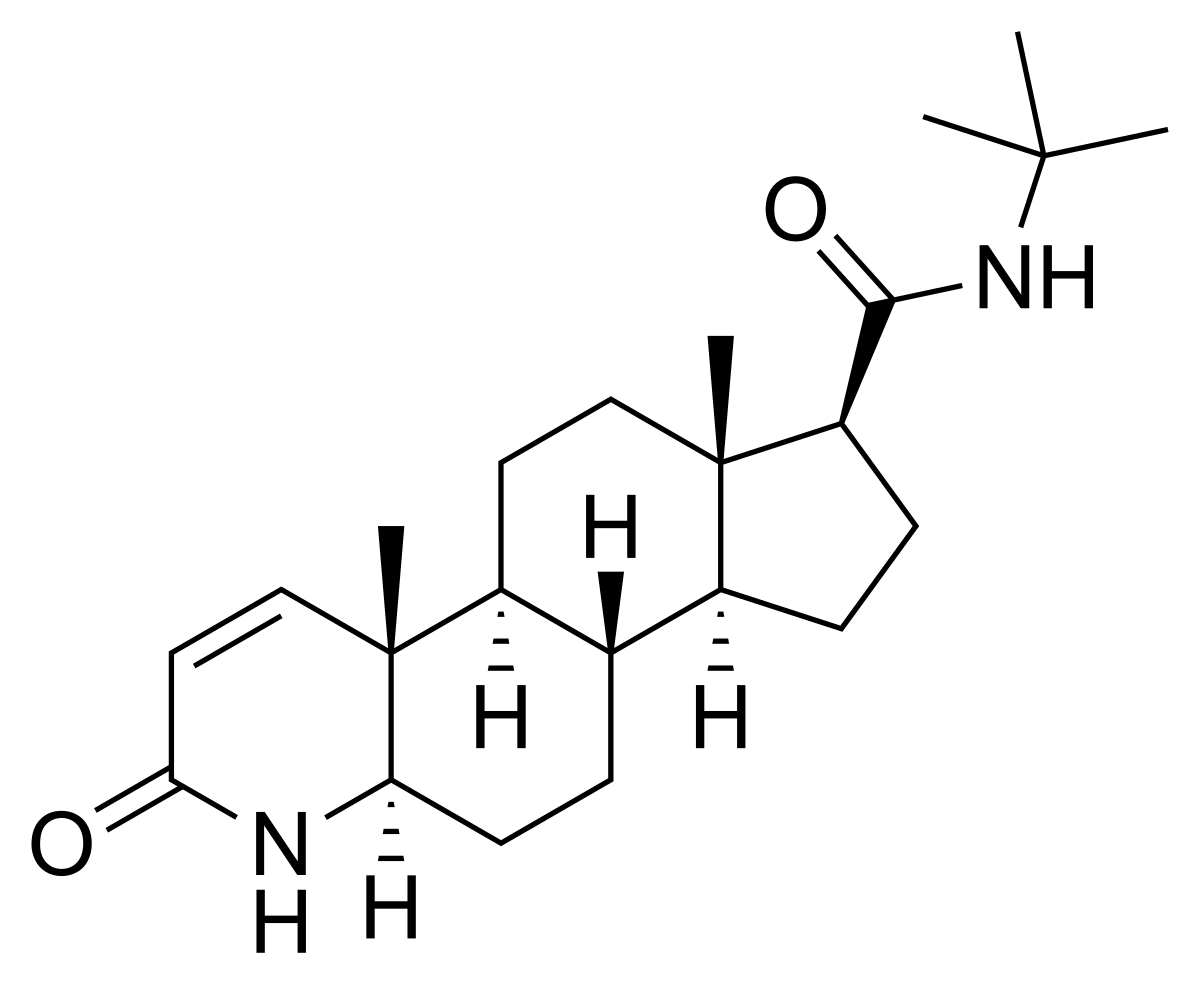
A 2019 Reuters investigation showed that Merck found evidence of persistent side effects in their original clinical trials and did not disclose it in their warning label.[82][83] They uncovered court filings which demonstrated that Merck misrepresented Propecia's safety record following clinical trials in the mid-late 1990's.[82]
In one deposition, Charlotte Merritt, who oversaw regulatory activity for Propecia, acknowledged that Merck changed Propecia's label for sexual adverse events in 2002, four years after Propecia hit the market. The label changed from “resolution occurred in all men who discontinued therapy with Propecia ” to “resolution occurred in men who discontinued therapy with Propecia”. Merritt testified that Merck eliminated the word “all” due to evidence from the clinical trials of adverse events that did not resolve following discontinuation of use.[83] In another deposition, Paul Howes, the head of marketing for Propecia acknowledged that Merck was aware that warnings of sexual side effects, particularly persistent to permanent side effects, would have a devastating impact on sales.[83]
 en.m.wikipedia.org
en.m.wikipedia.org
In one deposition, Charlotte Merritt, who oversaw regulatory activity for Propecia, acknowledged that Merck changed Propecia's label for sexual adverse events in 2002, four years after Propecia hit the market. The label changed from “resolution occurred in all men who discontinued therapy with Propecia ” to “resolution occurred in men who discontinued therapy with Propecia”. Merritt testified that Merck eliminated the word “all” due to evidence from the clinical trials of adverse events that did not resolve following discontinuation of use.[83] In another deposition, Paul Howes, the head of marketing for Propecia acknowledged that Merck was aware that warnings of sexual side effects, particularly persistent to permanent side effects, would have a devastating impact on sales.[83]