Informationcapitali
Goonmaster 4000.How to LDAR like a KING? darktriad
- Joined
- Feb 14, 2022
- Posts
- 3,489
- Reputation
- 4,035
Quite some time I've been suspicious were missing a very important pathway activation for bone mass upregulation and these articles I've just read through contain a lot of the missing puzzles to bone development and final size of bones.
First off though let me compile my findings.
Irregular bones such as the ones comprising the zygomatic bone, maxilla, jaw, etc.. all have outgrowth that is dependent intramembraneous ossification. Meaning that myschemial stem cells (MSC); the progenitors to bone laying osteoblasts undergo a direct conversion to osteoblastic lineages with correct osteoblastic factors being expressed. (Runx2, bmp,osteox, etc..)
This is important cause it let's us know that the factors that induce outgrowth of irregular bones do not overlap with and are antagonistic to Limb lengthening factors that look for MSC commitments to (chondrogenic) lineages.
So if we want to look for way to increase the size of (maxilla, zygomatic process, zygomatic bone,)we should increase MSC commitments to osteoblastic lineages.
Well I turned to research paper (Effects of Dlx2 overexpression on the genes associated with the maxillary process in the early mouse embryo) to get a better understanding on how a gene (DLX2) known for playing a role in growth guidance of the maxillary primordia and the skeletal elements that derive from this through being a potent signaller to (MSC) to differentiate into osteoblasts.
Well the findings of the upregulation of DLX2 on the lab rats ends up being unexpectedly suppressive to the full width and half-width of the maxilla of the mice.
Illustration below.
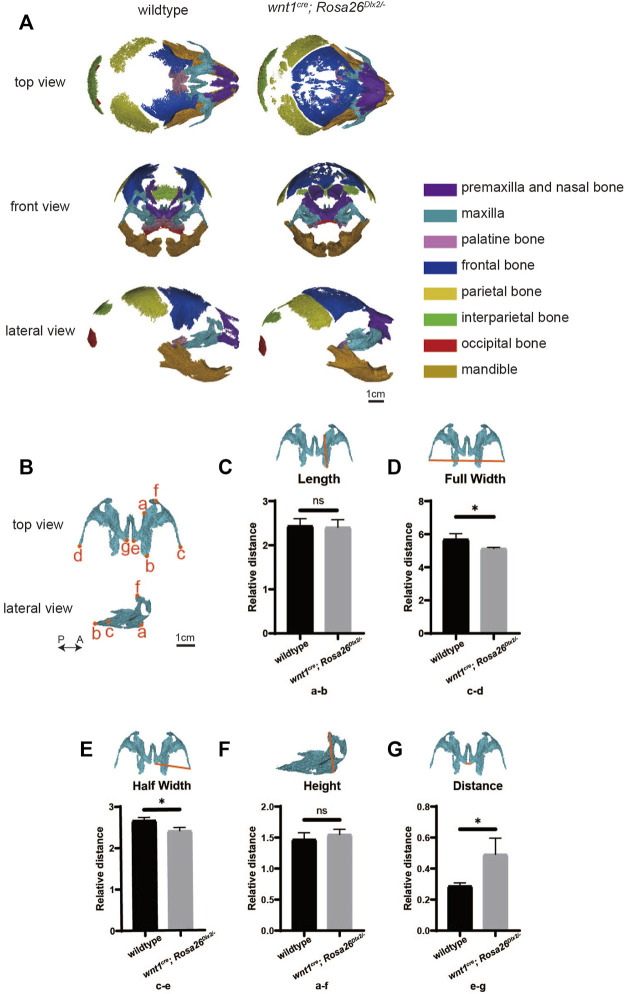
The study determind that the reason for the reduction in maxilla width was due to DLX2 being an extremely potent osteoblastic lineages promoter.
Leading to
1.premature differentation of (MSC)
2. A smaller pooled of undifferentiated msc
So what we can infer is that in order to create the necessary environment to promote bigger bones, we need to activate (MSC) proliferation pathways.
Another study backing my view on this is researchers hinting at the cluster of (MSC) of small faced dogs (brachycephalic dogs) that are destined to become zygoma being much greater in proportion to the maxilla when compared to a non-brachycephalic breed.
" Thus among brachycephalic dogs, the zygoma is relatively larger and the maxilla relatively smaller than one might expect from sampling non‐brachycephalic breed dogs. It is tempting to speculate that this observation is hinting at an imbalance of progenitor cells destined to contribute to formation of the maxilla and the zygoma."
One last study solidifying the view that MSC proliferation is more important the pi3k pathway activation is the study below.
Six2 expression has been associated with increased mesenchymal cell proliferation in the developing head and renal system [48, 49, 74]. Recent results indicate that Six2 mRNA and protein levels are highest in palatal tissues during the period of initial palatal shelf outgrowth and suggest that later spatiotemporal expression patterns are responsible for local increases in mesenchymal cell proliferation [48]. It is possible that genetic variation under our candidate region leads to a change in the timing, location, or level of mesenchymal precursor cell populations.
A change in proliferation within either the maxillary or zygomatic mesenchymal condensations may result in size variation of that condensation and the resulting bones.
What we can infer by the findings of the presented studies is that if we want larger bones (zygomatic arch, wrists, etc..) we need to increase MSC condensation sizes prior to them converting to preosteoblasts.
Problem is since minimal research has been done for identification of substances that increase MSC proliferation. We don't know of any natural substances ( like alpha gpc for somostatin inhibition), peptides, or agonists that can get us the results we want.
Your welcome to try finding a substance that can allow us to upregulate MSC proliferation using this extensive chemical database of all known substances affecting MSC that I linked below.
Identifying the correct substance
1.easily accessible for purchase
2.has research articles that validate its function in increasing (MSC) proliferation.
3. Able to be be injected or orally consumed
First off though let me compile my findings.
Irregular bones such as the ones comprising the zygomatic bone, maxilla, jaw, etc.. all have outgrowth that is dependent intramembraneous ossification. Meaning that myschemial stem cells (MSC); the progenitors to bone laying osteoblasts undergo a direct conversion to osteoblastic lineages with correct osteoblastic factors being expressed. (Runx2, bmp,osteox, etc..)
This is important cause it let's us know that the factors that induce outgrowth of irregular bones do not overlap with and are antagonistic to Limb lengthening factors that look for MSC commitments to (chondrogenic) lineages.
So if we want to look for way to increase the size of (maxilla, zygomatic process, zygomatic bone,)we should increase MSC commitments to osteoblastic lineages.
Well I turned to research paper (Effects of Dlx2 overexpression on the genes associated with the maxillary process in the early mouse embryo) to get a better understanding on how a gene (DLX2) known for playing a role in growth guidance of the maxillary primordia and the skeletal elements that derive from this through being a potent signaller to (MSC) to differentiate into osteoblasts.
Well the findings of the upregulation of DLX2 on the lab rats ends up being unexpectedly suppressive to the full width and half-width of the maxilla of the mice.
Illustration below.

The study determind that the reason for the reduction in maxilla width was due to DLX2 being an extremely potent osteoblastic lineages promoter.
Leading to
1.premature differentation of (MSC)
2. A smaller pooled of undifferentiated msc
So what we can infer is that in order to create the necessary environment to promote bigger bones, we need to activate (MSC) proliferation pathways.
Another study backing my view on this is researchers hinting at the cluster of (MSC) of small faced dogs (brachycephalic dogs) that are destined to become zygoma being much greater in proportion to the maxilla when compared to a non-brachycephalic breed.
" Thus among brachycephalic dogs, the zygoma is relatively larger and the maxilla relatively smaller than one might expect from sampling non‐brachycephalic breed dogs. It is tempting to speculate that this observation is hinting at an imbalance of progenitor cells destined to contribute to formation of the maxilla and the zygoma."
One last study solidifying the view that MSC proliferation is more important the pi3k pathway activation is the study below.
Six2 expression has been associated with increased mesenchymal cell proliferation in the developing head and renal system [48, 49, 74]. Recent results indicate that Six2 mRNA and protein levels are highest in palatal tissues during the period of initial palatal shelf outgrowth and suggest that later spatiotemporal expression patterns are responsible for local increases in mesenchymal cell proliferation [48]. It is possible that genetic variation under our candidate region leads to a change in the timing, location, or level of mesenchymal precursor cell populations.
A change in proliferation within either the maxillary or zygomatic mesenchymal condensations may result in size variation of that condensation and the resulting bones.
What we can infer by the findings of the presented studies is that if we want larger bones (zygomatic arch, wrists, etc..) we need to increase MSC condensation sizes prior to them converting to preosteoblasts.
Problem is since minimal research has been done for identification of substances that increase MSC proliferation. We don't know of any natural substances ( like alpha gpc for somostatin inhibition), peptides, or agonists that can get us the results we want.
Your welcome to try finding a substance that can allow us to upregulate MSC proliferation using this extensive chemical database of all known substances affecting MSC that I linked below.
Identifying the correct substance
1.easily accessible for purchase
2.has research articles that validate its function in increasing (MSC) proliferation.
3. Able to be be injected or orally consumed
Last edited: