TABLE OF CONTENTS
- Periosteal apposition myths
1) Wolff's law
2) subperiosteal hematoma ossification - Bonesmashing
1) Important Pathways, Receptors and signaling
2) How not to bonesmash
3) How to bonesmash
4) Routine - Bone building stack
1) Transdermal delivery system
2) how to home-brew transdermal solution
3) All pharmaceutical options - How to use steroids
Firstly a big shoutout to Wnba_young_bitch for helping me create the protocol.
Periosteal Apposition Myths
1) Wolff's Law
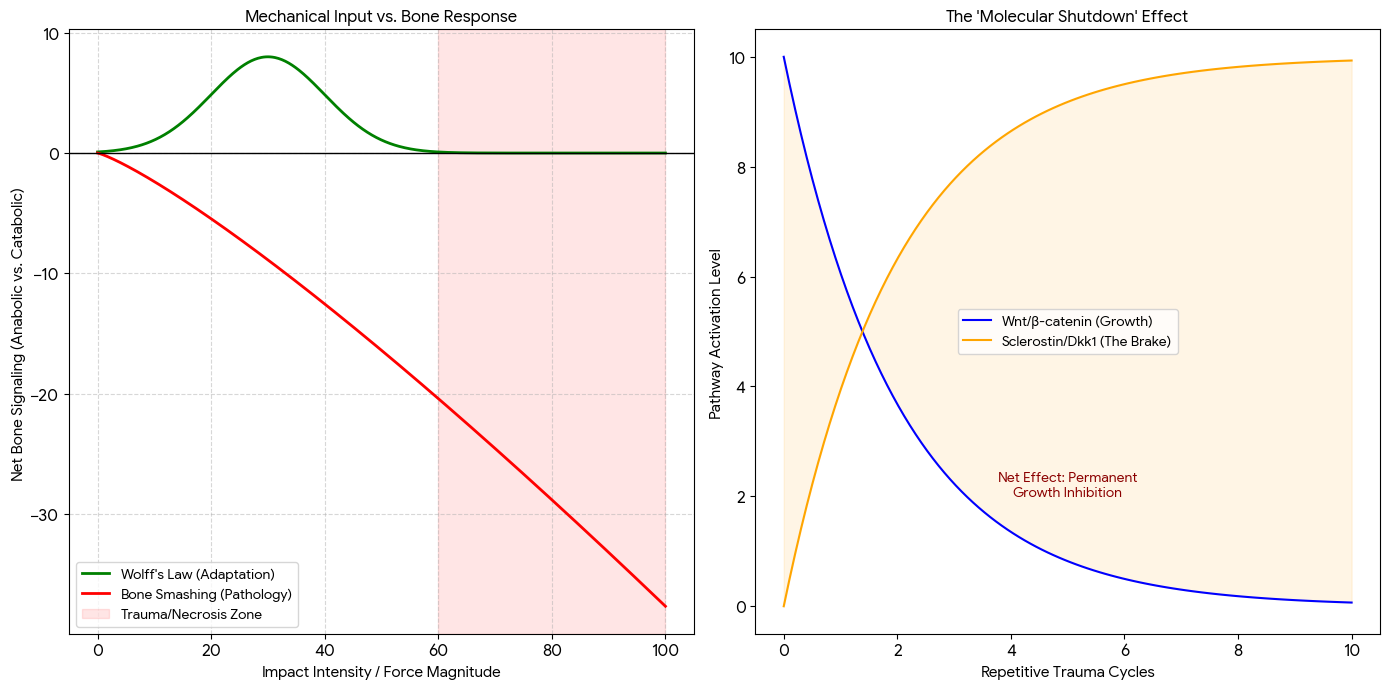
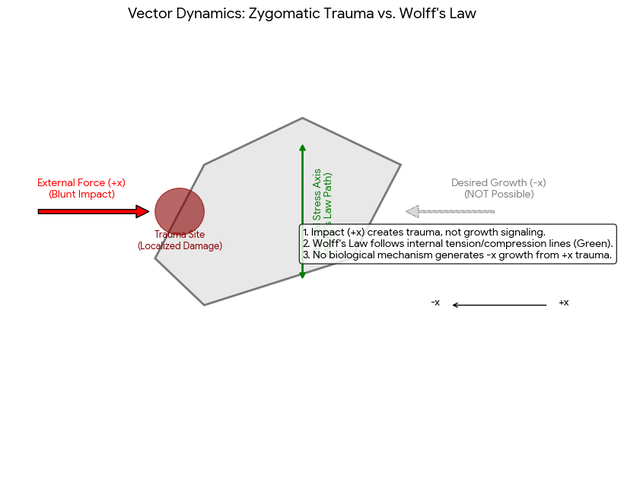
The misconception: Wolff's law states that a bone will adapt via bone density along with some minor shape changes under constant load over time, not from hitting the bone randomly, While Wolff’s Law explains how bones adapt to functional mechanical loads (like walking or lifting weights), it does not apply to blunt force trauma like bonesmashing. Cyclic stresses like chewing or orthodontics can subtly remodel them as per Wolff's Law, as seen in the functional matrix theory where soft tissues guide skeletal growth. we cannot strike the bone in +x direction and expect growth in -x direction. For example orthodontic devices exert constant force for several months for bone re-modeling (not extra bone growth) to change the shape of the existing bone (Wolff's law requires mechanotransduction—the process where cells detect tiny fluid movements within the bone caused by normal bending and compression. Blunt trauma in the (+x direction) . The body replaces the soft bridge with brittle, unorganized bone (woven bone), which often feels lumpy and lacks the strength of normal lamellar bone, and is not pronounced enough to appeal for aesthetics. Fibrocartilaginous Callus: The body builds a soft bridge to stabilize the trauma site from bonesmashing, The woven bone usually gets reabsorbed from the RANKL pathway which I will explain in the next section 'Subperiosteal hematoma ossification'.
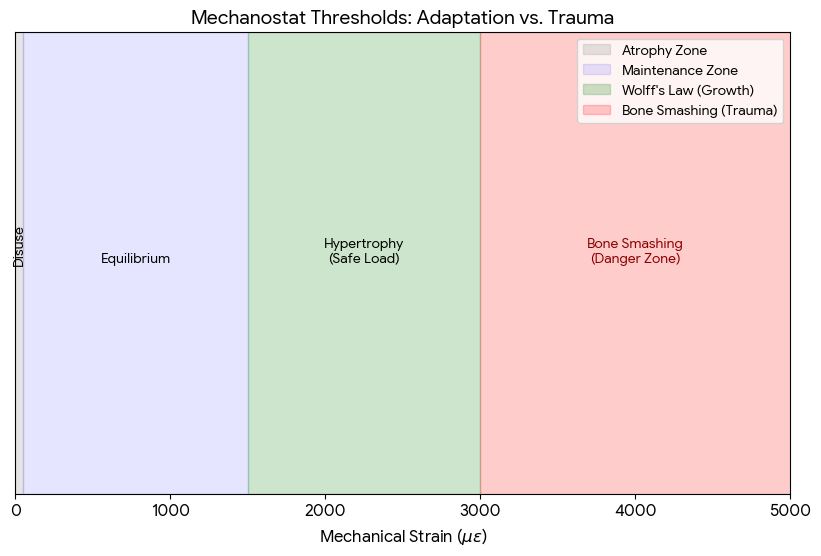
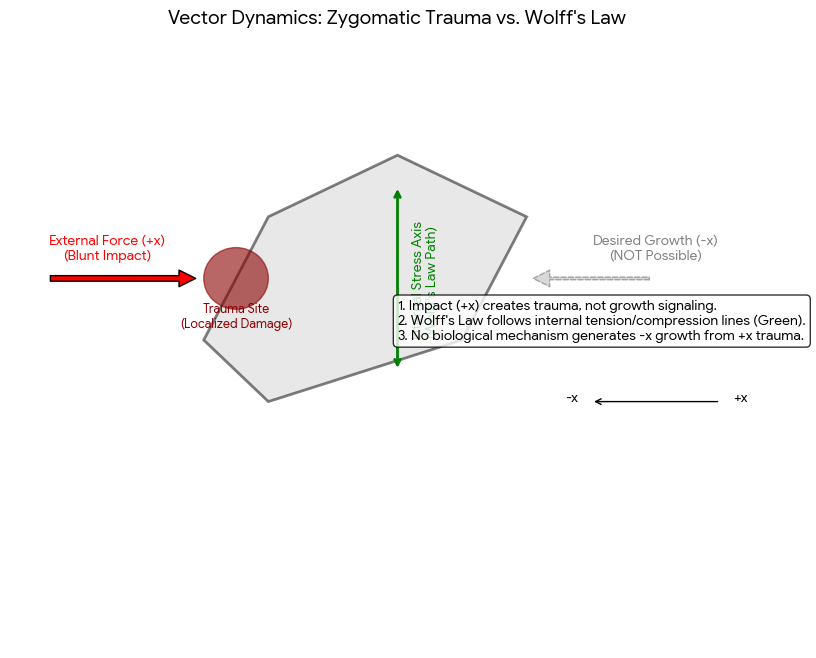
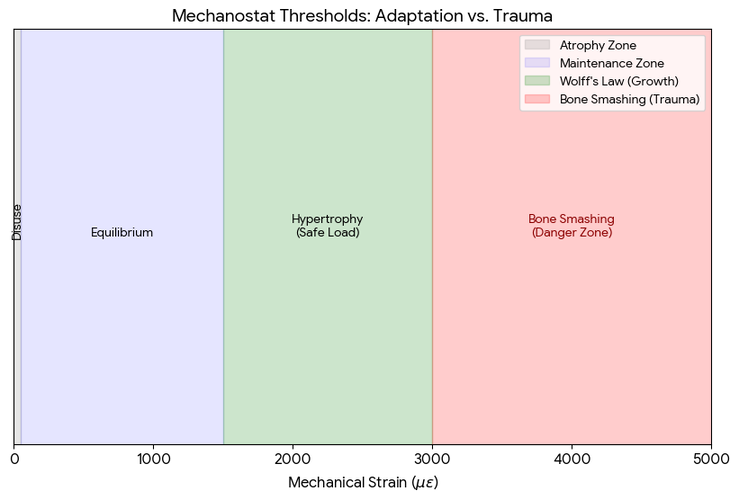
Diagram: as we can see from the diagram above (1) that the strain needs to be at a low dose between 1500-3000με of mechanical strain over a prolonged period of time (months). Strains in this range trigger osteoblasts to add bone mass (not necessarily bone volume) in an organized, functional way.
2) Subperiosteal hematoma ossification
What is it/theoretical basis: The theory begins with a high-velocity impact (via a hammer) to a bone like the zygoma (cheekbone) or chin (jaw). The impact is intended to rupture the Sharpey’s fibers that tether the periosteum (the vascular membrane covering the bone) to the underlying cortical bone. This rupture breaks the subperiosteal micro vessels, leading to a localized accumulation of blood—a Subperiosteal Hematoma. Which calcifies (ossification) into new bone matrix on the existing periosteum.
Cytokines: IL-1, IL-6, and TNF-α are released to recruit immune cells.
Growth Factors: TGF-β (Transforming Growth Factor beta), VEGF (Vascular Endothelial Growth Factor), and BMPs (Bone Morphogenetic Proteins) are secreted from the damaged tissue and platelets.
These factors are meant to activate the SMAD and Wnt/β-catenin pathways, which tell the body to begin building bone immediately to seal the injury site.
HIF-1α (Hypoxia-Inducible Factor): Because the hematoma cuts off the direct oxygen supply to the bone surface, the body expresses the HIF-1α gene.
Osteogenesis: In this theory, HIF-1α is the master switch that upregulates Runx2 and Osterix—the primary transcription factors that turn Mesenchymal Stem Cells (MSCs) in the periosteum's cambium layer into Osteoblasts (bone-building cells).
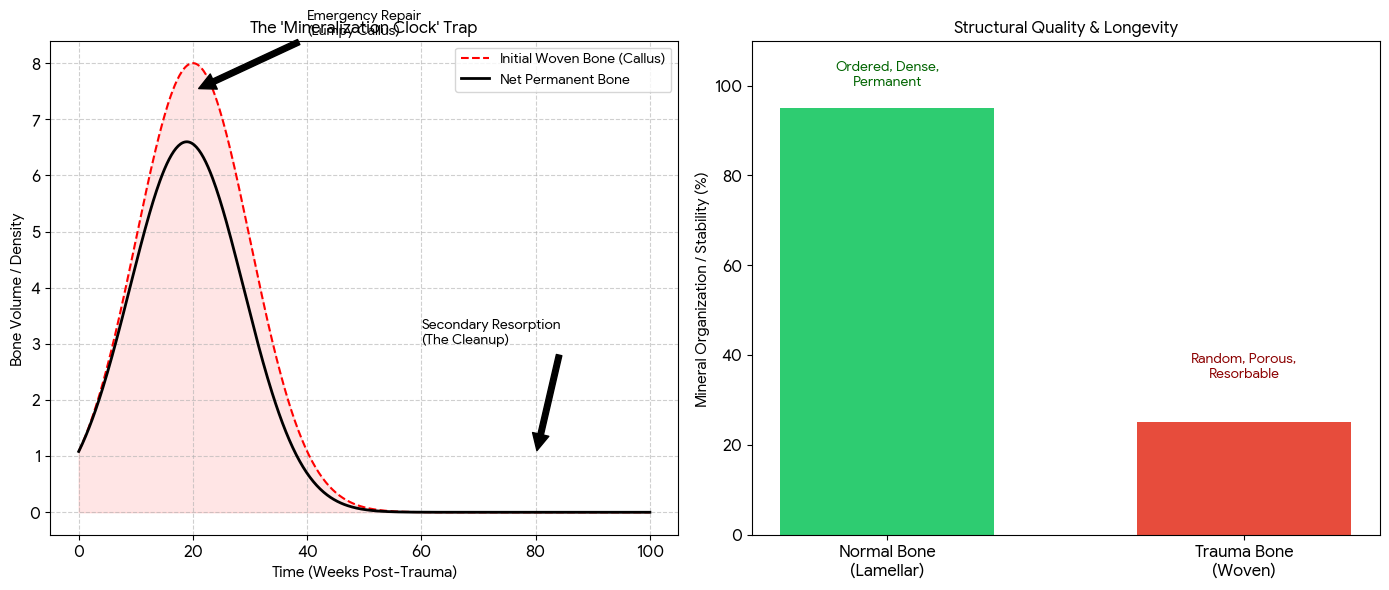
According to the theory, the Mineralization Clock (explained by jordan wood) begins. The body cannot wait to build slow, high-quality bone; it must stabilize the area.
Intramembranous Ossification: The activated osteoblasts lay down a collagen matrix directly into the hematoma.
Woven Bone: This matrix is quickly mineralized into Woven Bone. This is described by proponents as a callus that adds physical volume to the bone's exterior.
Streaming Potentials: Proponents argue that the pressure from the hematoma generates piezoelectric charges (electrical signals) in the bone’s hydroxyapatite crystals. These signals lead to permanent, new bone formation.
in depth explanation of the 4 key features of the theory and how they are proved wrong
enjoy!
enjoy!
1. inflammatory environment: When a subperiosteal hematoma occurs as a result of an injury or a direct blow to the bone the blood vessels connecting the periosteum and the bone rupture leading to blood accumulation and the spread of red blood cells. After some time these cells coagulate and cause the release of a group of inflammatory factors from the blood components such as platelets macrophages and other immune cells. These factors include cytokines such as IL-1β IL-6 TNF-α as well as chemokines like MCP-1 and growth factors such as TGF-β VEGF BMPs and Cox2. inflammatory cytokines help attract mesenchymal stem cells (MSCs) from the bone marrow and the inner layer of the periosteum which is very rich in MSCs to the injury site, These stem cells migrate toward the affected area due to chemokine signals and are activated by cytokines and inflammatory mediators like IL-6 and TGF-β, Upon arrival the stem cells begin differentiating into osteoblasts thanks to stimulation by growth factors such as BMPs and TGF-β leading to the secretion of the initial bone matrix, activating signaling pathways such as SMAD MAPK and JAK/STAT
Flaw #1 The TNF-α Paradox The theory cites TNF-α as a growth factor. While low-level, transient TNF-α is a chemotactic signal for MSCs, high-level or chronic TNF-α (caused by repeated smashing) is a potent inhibitor of bone formation. Chronic TNF-α activates the NF-κB pathway, which directly suppresses Runx2 (An important transcription factor for bone) TNF-α induces the expression of Dickkopf-1 (Dkk-1) and Sclerostin (SOST). These are the "master brakes" of the Wnt/β-catenin pathway. Without Wnt signaling, MSCs cannot complete differentiation into osteoblasts; they either remain dormant or undergo adipogenesis (turning into fat cells) instead of bone.
Flaw #2 Cytokine-Induced Osteoclastogenesis The exact cytokines listed (IL-1β, IL-6, and TNF-α) are the primary drivers of bone resorption. These cytokines stimulate osteoblasts and T-cells to overexpress RANKL (Receptor Activator of Nuclear Factor κB Ligand) while simultaneously suppressing Osteoprotegerin (OPG) A high RANKL/OPG ratio activates Osteoclasts. In a subperiosteal hematoma environment, this is useless and infact counter productive since the top priority is reabsorption of useless bone volume. Because the trauma is external and blunt, the osteoclastic resorption of the damaged underlying cortical bone often outpaces the disorganized deposition of new woven bone, leading to net bone loss (osteolysis).
Flaw #3 MAPK Pathway Overload and Senescence The theory mentions the MAPK (Mitogen-Activated Protein Kinase) pathway as a positive. But in reality excessive mechanical trauma and high-dose oxidative stress (from the iron in the heme of the hematoma) cause p38 MAPK over-activation. This triggers Cellular Senescence in the very MSCs you are trying to recruit. Senescent MSCs develop a SASP (Senescence-Associated Secretory Phenotype), where they pump out more inflammatory cytokines, creating a feedback loop that prevents mineralization and leads to fibrotic scarring instead of ossification.
Flaw #4 The Heme-Toxic Microenvironment The theory treats the pool of blood as a nutrient source. In reality, a large subperiosteal hematoma is cytotoxic. As red blood cells in the hematoma lyse, they release hemin and free iron. Free iron triggers the Fenton Reaction, producing Reactive Oxygen Species (ROS). High ROS levels inhibit SMAD signaling (the pathway mentioned in the theory), effectively blunting the effect of any BMPs present. Instead of bone, the body often produces fibrovascular granulation tissue—a permanent, soft-tissue lump that feels like bone but has zero structural or aesthetic integrity, this is basically scar tissue.
electrical potential: Bone has piezoelectric properties meaning it generates electrical signals when subjected to stress or strain. When blood accumulates under the periosteum it changes the mechanical stress pattern on the bone generating a local electrical potential in the affected area. This electrical potential acts as a signal that stimulates osteocytes and mesenchymal stem cells (MSCs) in the area. The stem cells respond to these electrical signals by migrating to the injury site via integrin receptors on their surface and differentiating into osteoblasts. Once these cells reach the site they begin secreting bone matrix made of collagen and calcium salts contributing to the formation of new bone. Furthermore the change in electrical stress activates signaling pathways such as the calcium pathway and MAPK leading to the opening of calcium channels in bone cells. Simply put this theory is based on the idea that the change in electrical potential tricks your body into believing that a fracture has occurred in the bone when in reality nothing has happened to it
Flaw #1 The Streaming Potential Paradox The theory claims that a hematoma tricks the bone via electrical potential. In reality, bone cells (osteocytes) are tuned to Dynamic Streaming Potentials, not static pressure. Piezoelectricity in bone occurs when the Hydroxyapatite crystals are deformed and then released, causing fluid to flow through the lacunocanalicular system. This fluid flow creates a hypothetical streaming potential. A subperiosteal hematoma creates Static Hydrostatic Pressure. Static pressure does not move fluid; it traps it. Without fluid oscillation, there is no consistent streaming potential. To the osteocyte, a hematoma is not a growth signal.
Flaw #2 The Voltage-Gated Calcium Channel (VGCC) burnout The theory mentions the Calcium Pathway and MAPK. While true that electrical signals open calcium channels, "Bone Smashing" triggers Pathological Hypercalcemia at the cellular level. Sudden and massive blunt trauma causes an uncontrolled influx of Ca2+ through L-type Voltage-Gated Calcium Channels and Stretch-Activated Channels (SACs) Instead of a healing signal this massive calcium spike triggers the Calpain-Caspase Pathway, leading to Programmed Cell Death (Apoptosis) of the very MSCs and osteocytes you are trying to trick. The trick doesn't lead to growth; it leads to a Necrotic Zone where no bone can physically form.
Flaw #3 Dielectric Shielding The theory suggests the blood creates the electrical signal. Physics dictates the opposite: Blood is a highly conductive, ionic fluid that acts as a Faraday Shield. For a piezoelectric signal to reach a cell, it must travel through the bone matrix. A subperiosteal hematoma is essentially a pool of electrolytes (sodium, potassium, chloride). This ionic pool dissipates (shorts out) any localized electrical potentials generated by the bone. Instead of tricking the body into thinking there is a fracture, the hematoma electrically insulates the bone surface from the mechanical strains of normal life, potentially leading to Local Disuse Atrophy (bone thinning) under the site of the hit.
severe oxygen deprivation: When the bone is exposed to a direct impact the blood vessels rupture leading to blood clotting which creates a highly hypoxic environment. Recent research shows that hypoxia plays a crucial and essential role in bone formation. The rupture of blood vessels cuts off the oxygen supply to the bone which acts as a trigger for the body to increase the expression of a gene called HIF-1α (Hypoxia Inducible Factor-1 alpha).This protein or gene is very important for osteogenic differentiation through multiple pathways. First by increasing the expression of VEGF second by enhancing the activity of the Wnt/β-catenin signaling pathway and also by upregulating BMPs. Moreover it inhibits the activity of Twist2 which leads to an increase in the gene expression of Runx2 and Osterix. Simply the hypoxic environment is ideal for the differentiation of stem cells attracted from the inner layer of the periosteum by inflammatory factors as it drives them to differentiate into osteogenic cells and form a mature and complete bone matrix at the injury site (the site of the hematoma)
Flaw #1 The Metabolic Dead End The theory claims hypoxia is ideal and essential for differentiation. This is a Bioenergetic Fallacy. Converting a Mesenchymal Stem Cell (MSC) into an Osteoblast requires a massive increase in Mitochondrial Biogenesis and ATP production to synthesize collagen and pump calcium. Under the severe hypoxia of a subperiosteal hematoma, the Pasteur Effect kicks in. Cells switch to Anaerobic Glycolysis, producing lactic acid. This drops the local pH (acidosis). Low pH inhibits hydroxyapatite mineralization. Even if HIF-1α signals for a matrix, the acidic environment prevents that matrix from ever becoming mature and complete bone. You get a soft, unmineralized pre-bone that is quickly reabsorbed by the body.
Flaw #2 The HIF-1α double Pathway (Sox9 vs. Runx2) The theory claims HIF-1α upregulates Runx2 (Bone). In the reality of severe trauma, the opposite occurs via the Chondrogenic Switch. High levels of HIF-1α actually stabilize Sox9, the master transcription factor for Cartilage, while suppressing the late-stage maturation of Runx2. This is why fractures heal with a Soft Callus (Fibrocartilage) first. If you keep bonesmashing specifically to maintain a hematoma and maintaining hypoxia, the tissue never transitions to bone. You are essentially trying to build a jawline out of rubbery cartilage, which lacks the structural rigidity of the craniofacial skeleton and eventually undergoes fibrotic degradation (reabsorption).
Flaw #3 The VEGF Vessel Trap (Angiogenic-Osteogenic Uncoupling) The theory highlights VEGF (Vascular Endothelial Growth Factor) as a positive. VEGF needs a stable gradient to grow organized blood vessels (Angiogenesis). Repeated trauma ruins the delicate new sprouts (capillary buds) as they try to enter the hematoma. This creates Non-Union conditions. You have high VEGF but zero functional blood flow. Without oxygenated blood to bring in nutrients and clear out metabolic waste (CO2/Lactic Acid), the MSCs enter Autophagy and die. HIF-1α cannot save a cell that has no substrate to build with.
Flaw #4 The Twist2/Runx2 Misinterpretation The theory suggests inhibiting Twist2 leads to mature bone. Twist2 is a gatekeeper that prevents premature differentiation. When trauma forces Twist2 down and Runx2 up in a high-stress, low-oxygen environment, it leads to Osteoblast Senescence. The cells burn out. They produce a burst of poor-quality Woven Bone (the Mineralization Clock Trap explained by Jordan Wood) and then die. Because they are not supported by a vascular network, (Avascular Necrosis) and is targeted for immediate removal by the body's immune system.
Tensile force caused by the hematoma: This hypothesis may be the most logical one since many scientists have stated that it is the most reasonable explanation for the ossification of a subperiosteal hematoma. This theory suggests that the hematoma lifts the periosteum away from the bone layer which activates the MSCs located in the inner layer of the periosteum (the cambium layer) through their mechanoreceptors such as primary cilia integrins and purinergic receptors. Researchers supported this hypothesis based on an in vitro study where stem cells extracted from the inner layer of the periosteum were subjected to tensile force (similar to what cells might experience during a hematoma) and this indeed led to an increased gene expression of Runx2 Osterix and BMP which are crucial genes and proteins responsible for driving MSCs to differentiate into osteogenic cells
Flaw #1 The Signal Saturation and Deciliation Trap. The theory assumes that more force equals more activation. Biology dictates the opposite via Ciliary Resorption. The primary cilium is an exquisitely sensitive antenna designed to detect low-level oscillatory fluid flow. High-magnitude blunt trauma (smashing) and the resulting massive hydraulic pressure of a hematoma exceed the structural limits of the cilium. This triggers AURKA (Aurora A kinase), leading to Deciliation—the retraction or shedding of the cilium. Without its antenna, the mesenchymal stem cell (MSC) becomes blind to any osteogenic signaling. Instead of differentiating, the deciliated cell enters Cell Cycle Arrest, creating a permanent, non-bony scar.
Flaw #2 Integrin Overloading and Anoikis-like Stress The theory cites integrins as the migration trigger Integrins require a stable extracellular matrix (ECM) to provide traction for the cell to move and differentiate. Blunt trauma and the resulting hematoma physically shatter the ECM and the Sharpey's fibers. This is Biomechanical Decoupling. The integrins have nothing to grip on to. When MSCs lose their specific attachment to the matrix due to trauma, they trigger Anoikis (detachment-induced apoptosis). You aren't recruiting cells; you are creating a necrotic area.
Flaw #3 The Hippo/YAP Pathway Reversal The most critical factor in mechanobiology is the Hippo/YAP pathway Osteogenic differentiation requires YAP/TAZ to translocate into the nucleus. This only happens when cells are on a stiff, stable substrate. A hematoma is a liquid/gelatinous substrate. MSCs on soft substrates (like a pool of blood) are biologically forced into the Adipogenic (fat-forming) or Chondrogenic (cartilage) lineage. the biological pathways produce subcutaneous fat deposits or soft fibrous tissue.
Flaw #4 The Purinergic Burnout & ATP Excitotoxicity The theory lists purinergic receptors (which detect ATP) as a trigger Trauma causes a massive, localized release of extracellular ATP from crushed cells. Chronic high-dose ATP exposure causes Purinergic Desensitization. The receptors shut down to prevent cell death. This mutes the calcium signaling mentioned in Theory #2. The MSCs lose their ability to respond to any further mechanical cues, leading to a localized zone of Metabolic Hibernation.
Bonesmashing
1) Important Pathways, Receptors and signaling (not all of them)
Wnt/β-Catenin Signaling, GSK-3β, Sclerostin (SOST), DKK-1 — Core osteogenic switch controlling osteoblast differentiation and bone mass.
Mechanotransduction Axis — Piezo1, Piezo2, TRPV4, TREK-1, Integrins, FAK, Fluid Shear Stress, Prostaglandin E2 (PGE2), Prostaglandin E1 (PGE2), EP4 Receptors EP2 receptor.
Androgen Receptor Signaling, SHBG Regulation — Skeletal response to testosterone and DHT.
Growth Hormone (GH) and IGF-1 Axis — Osteoblast proliferation and periosteal expansion.
Estrogen Receptor Signaling (ERα, ERβ) — Suppression of resorption and regulation of RANKL/OPG balance.
BMP Signaling (BMP-2, BMP-4, BMP-7) — Osteoinduction and new bone formation.
TGF-β / TGF-α / SMAD Signaling — Collagen synthesis, extracellular matrix deposition, bone repair and remodeling.
Angiogenesis Signaling — VEGF, Nitric Oxide, HIF-1α vascular coupling to osteogenesis.
cAMP Second Messenger System and PDE Enzymes — Amplification and regulation of osteogenic signaling.
Neuro-Osteogenic Coupling — CGRP, Substance P neuropeptide signaling.
Sympathetic Nervous System Signaling — β2-Adrenergic receptors regulating bone turnover.
RANKL / OPG System — Osteoclast activation vs resorption inhibition.
Calcitonin Receptor Signaling — Direct suppression of osteoclast activity.
Epigenetic Regulation — HDAC activity, Runx2, Osterix transcriptional control.
Notch Signaling — Skeletal stem cell maintenance and differentiation timing.
Hippo Pathway, YAP / TAZ Signaling — Regulation of proliferation and skeletal expansion.
PTHrP Signaling — Maintenance of proliferative osteoprogenitor state.
Lacuno-Canalicular Communication — Connexin-43 gap junction signaling.
mTOR Pathway — Osteoblast growth, protein synthesis, matrix production.
AMPK Pathway — Mitochondrial health and osteocyte survival.
MAPK Signaling — ERK and p38 pathways driving osteogenic gene expression.
Hedgehog Signaling — Indian Hedgehog (IHH), Sonic Hedgehog (SHH) craniofacial development.
Calcium-Sensing Receptor (CaSR) Signaling — Mineral deposition and calcium homeostasis.
FGF / FGFR Signaling — Osteoblast proliferation, angiogenesis, craniofacial growth.
Osteocyte Autophagy and Apoptosis Pathways — Maintenance and remodeling triggers.
Reactive Oxygen Species (ROS) Signaling — Redox regulation of osteogenesis.
Circadian Clock Genes — BMAL1, CLOCK, PER, CRY bone turnover rhythms.
Osteoimmunology — IL-6, TNF-α, IL-1β inflammatory regulation of remodeling.
Matrix Remodeling Enzymes — MMP-2, MMP-9, Cathepsin K structural reshaping.
Periosteal Stem Cell Niche — Mechanosensitive progenitors driving appositional growth.
Hypoxia Signaling — HIF-1α oxygen sensing and angiogenesis.
Lactate Signaling — Metabolic regulation of osteoblast differentiation and vascular growth.
Wnt/β-catenin signaling is the primary regulator of osteoblast differentiation and bone mass, with GSK-3β degrading β-catenin to suppress signaling, Sclerostin acting as a major extracellular brake on Wnt activity, and DKK-1 preventing Wnt ligands from binding their receptors. Bone mechanotransduction converts physical load into biochemical signals through Piezo1 and Piezo2 ion channels, TRPV4 and TREK-1 mechanically gated calcium channels, integrins and focal adhesion kinase signaling, fluid shear stress within the lacuno-canalicular network, and prostaglandin E2 acting through EP4 receptors. Hormonal regulation includes androgen receptor density determining skeletal responsiveness to testosterone and DHT, SHBG regulating free hormone availability, growth hormone and IGF-1 driving osteoblast proliferation and periosteal expansion, and estrogen receptor signaling suppressing bone resorption while regulating the RANKL/OPG balance. Osteoinduction is driven by BMP signaling including BMP-2, BMP-4, and BMP-7, alongside the TGF-β family including TGF-β1, TGF-β2, TGF-β3, and TGF-α which regulate collagen synthesis, extracellular matrix deposition, and bone repair. Angiogenesis and vascular coupling are mediated by VEGF, nitric oxide, and HIF-1α to supply growing bone with blood vessels. cAMP functions as a central second messenger amplifying osteogenic signaling while phosphodiesterase enzymes regulate its degradation. Neuro-osteogenic coupling includes CGRP and Substance P stimulating osteoblast activity and sympathetic nervous system β2-adrenergic receptors influencing bone turnover. Bone remodeling balance is governed by the RANKL/OPG system, with RANKL activating osteoclasts, osteoprotegerin acting as a decoy receptor to prevent resorption, and calcitonin directly inhibiting osteoclast activity. Epigenetic regulation involves HDAC activity controlling expression of Runx2 and Osterix, the master osteogenic transcription factors, while Notch signaling maintains skeletal stem cell pools and regulates differentiation timing. The Hippo pathway restrains tissue growth, and when inhibited allows YAP and TAZ to promote proliferation and skeletal expansion. PTHrP signaling maintains osteoprogenitor cells in a proliferative state, extending the window for bone formation. Lacuno-canalicular communication is coordinated through Connexin-43 gap junctions between osteocytes and osteoblasts. Cellular energy sensing is controlled by mTOR promoting osteoblast growth and matrix synthesis and AMPK supporting mitochondrial health and osteocyte survival. MAPK pathways including p38 and ERK translate mechanical and chemical signals into osteogenic gene expression. Hedgehog signaling through Indian Hedgehog and Sonic Hedgehog regulates osteoblast formation, chondrocyte maturation, and craniofacial morphogenesis. Calcium signaling through the calcium-sensing receptor regulates mineral deposition and osteoblast differentiation. FGF and FGFR signaling stimulates osteoblast proliferation, angiogenesis, and craniofacial growth. Osteocyte survival depends on autophagy and apoptosis pathways that trigger remodeling when needed. Reactive oxygen species act as signaling molecules at low levels to stimulate osteogenesis but promote bone loss when excessive. Circadian clock genes including BMAL1, CLOCK, PER, and CRY regulate daily bone turnover cycles. Osteoimmunology links bone to immune signaling through cytokines such as IL-6, TNF-α, and IL-1β which regulate remodeling and inflammation. Extracellular matrix remodeling requires matrix metalloproteinases MMP-2 and MMP-9 and Cathepsin K to enable structural reorganization. The periosteal stem cell niche contains mechanosensitive progenitors responsible for bone widening and surface apposition. Hypoxia signaling through HIF-1α and metabolic lactate signaling promote angiogenesis and osteoblast differentiation, completing the integrated network governing skeletal expansion and bone remodeling.
2) How not to bonesmash
Hit with a hammer for 30min
3) How to bonesmash
Bonesmashing refers to the deliberate application of controlled mechanical trauma or Low-impact high frequency loading to facial bones (Zygos, chin, supraorbital ridge, and gonion) to induce hypertrophy; in our case periosteum apposition. There are multiple pathways which come into play. The main factors were looking at are Wnt upregulation and sclerostin and Sost inhibition. Keep in mind that bonesmashing is not mechanical load, its a stimulus to the bone.
Bones have osteocytes which are cells that can convert mechanical stressors into cellular signals by activating sensors. When osteocytes sense a mechanical stressor they secrete nitric oxide and PGE2 (When you use your massage gun, the bone physically bends (micro-strain). This squeeze is what causes bone cells (osteocytes) to instantly flood the area with PGE2. PGE2 binds to EP4 receptors, which triggers the Wnt/β-catenin pathway; this is the primary driver of periosteum apposition. with the goal of blocking SOST and sclerostin. Lowering SOST and sclerostin results in more Wnt which is a pathway very anabolic for bone because it increases the amount of cells that produce bone and also suppresses bone resorption. Without SOST and sclerostin downregulation there is no bone growth from a mechanical stressor.
To fix recession and gain dimorphism, the goal is not to break the bone, but to achieve High-Frequency, Low-Magnitude (HFLM) loading. This triggers the Mechanostat, the internal biological sensor that dictates bone size.
The Physics of Vibration (30–50Hz): Bone is a poroelastic material. When you apply a massage gun at 30–50Hz (vibrations per second), you create a phenomenon called Interstitial Fluid Flow. Lacunocanalicular System (LCS): Inside the bone is a network of tiny tunnels. The vibration shakes the fluid inside these tunnels. Fluid Shear Stress: This moving fluid physically brushes against the membranes of Osteocytes (the architect cells), which is the primary signal for new growth.
Receptor Activation: The Piezo1 Gateway: At the cellular level, the massage gun targets Mechanosensitive Ion Channels, specifically Piezo1. The Action: Under the stress of the 30-50Hz vibration, the Piezo1 gate physically pulls open, allowing a surge of Calcium (Ca2+) into the cell. The Result: This calcium spike is the starting gun for the entire growth cascade. Without it, the pharmaceuticals in the stack have no mechanical direction on where to build.
The Trigeminal-Neurogenic Coupler: The face is uniquely innervated by the Trigeminal Nerve. CGRP Release: Vibration stimulates sensory nerve endings, which release Calcitonin Gene-Related Peptide (CGRP) and Substance P. Neuro-Osteogenesis: These neuropeptides act as potent local anabolics, telling the bone: "This area is under extreme load; increase volume here."
THE ROUTINE
This routine is engineered to synchronize the mechanical trigger (Piezo1) with the chemical signals (BMP-2/PGE2/AR) and the enzymatic locks (PDE4/AChE)
(T-Minus 60 minutes)
- Tadalafil (5mg): Dilates the microvasculature (angiogenesis) to ensure blood flow can pull the topical nutrients into the bone matrix.
- Pentoxifylline (PTX) (400mg): Lowers blood viscosity and begins the initial suppression of Sclerostin.
- Dipyridamole (75mg): Elevates local Adenosine, priming the A2A receptors for surface area expansion.
Apply the refined DMSO solution to the jawline, chin, and brow ridge.
- Fluvastatin (2%): Local BMP-2 induction for fixing recession.
- L-Carnitine (LCLT) (5%): Local Androgen Receptor (AR) upregulation to capture the 500mg Testosterone.
- Dinoprostone (PGE2): Direct EP4/EP2 agonists for forced widening.
- Misoprostol (200mcg): Provides the synthetic PGE2 signal to saturate the EP4 receptors before they are mechanically stimulated.
- WAY-316606: Neutralizes sFRP-1 so the Wnt receptors are unlocked.
Note: Some researchers use Occlusion (plastic wrap) for these 20 minutes to drive the DMSO deeper into the periosteum.
(The 30-Minute Session)
- Device: Massage Gun with a firm/flat head.
- Frequency: 30–50Hz (The Sweet Spot for Piezo1 and Interstitial Fluid Flow).
- Technique: Use moderate pressure. Focus on the mandibular angle, lower border of the jaw, and zygomatic arch.
(Immediately After)
- Lithium Orotate (10-20mg): Inhibits GSK-3β to prevent the "build" signal from being degraded.
- Donepezil (5mg): Inhibits AChE to stop the RANKL (bone-eating) response that usually follows heavy mechanical stress.
- Collagen (6000mg) + Vitamin D3: Provides new amino acids to fill the new space signaled by the vibration.
- Vitamin MK-4 (45mg): The Architect that cross-links the new collagen into a solid, masculine frame.
- Roflumilast (500mcg): The final blow to Sclerostin. It ensures the Wnt pathway stays open all night during the GH pulse.
Bone-building Stack
| Component | Research Goal |
|---|---|
| Fluvastatin | BMP-2 Induction |
| Misoprostol | PGE1 Signal |
| Dinoprostone | PGE2 Signal |
| L-Carnitine (LCLT) | AR Sensitizer |
| WAY-316606 | sFRP-1 Inhibitor |
| Lithium Orotate | GSK-3β Inhibitor |
- Mixing: Combine the powders in a dark glass dropper bottle. Add the 99.9% Pharmaceutical Grade DMSO. Shake until the solution is clear. If using crushed tablets (Fluvastatin), filter the sediment (binders/fillers) through a coffee filter or syringe filter.
- Skin Preparation: Thoroughly clean the jawline and brow with warm water. Avoid all soaps or oils before application, as DMSO will carry them into the tissue.
- The "Pre-Pump": Apply the gel after your 5mg Tadalafil has reached peak plasma (approx. 2 hours post-ingestion). The increased local blood flow will help "pull" the topical solution into the bone matrix.
- Loading Window: Apply the solution and wait 15–20 minutes for initial penetration. Some researchers use Occlusion (plastic wrap) over the jaw during this window to prevent evaporation and force deeper depth.
- The Kinetic Drive: Immediately follow with your 30-minute Massage Gun session (30-50Hz). The mechanical vibration acts as a "kinetic pump," driving the Fluvastatin and PGE2 through the soft tissue toward the bone's surface.
2) How to home-brew the transdermal solution
| Component | Concentration | Amount for 30ml | Preparation Note |
|---|---|---|---|
| Fluvastatin | 2% | 600 mg | Best used as pure powder; if using tablets, must be crushed and filtered. |
| Misoprostol | 0.5% | 150 mg | Extremely potent; requires high-precision milligram scale. |
| Dinoprostone | 0.5% | 150 mg | Highly sensitive to light and heat; add last. |
| L-Carnitine (LCLT) | 5% | 1500 mg | Large molecule; requires thorough shaking to dissolve. |
| WAY-316606 | 0.5% | 150 mg | Highly lipophilic; dissolves readily in DMSO. |
| Lithium Orotate | Trace | 50 mg | Acts as a local GSK-3β lock. |
- Preparation: Work in a clean, dry environment. Ensure all glass containers are sanitized and completely dry, as water can cause DMSO to generate heat and degrade the prostaglandins.
- Solvent Base: Measure 30ml of 99.9% DMSO into a glass beaker.
- The Solid Phase: Add the LCLT (1.5g) first, as it is the largest mass. Stir or shake until the liquid is clear. Follow with Fluvastatin (600mg) and Lithium Orotate (50mg).
- The High-Potency Phase: Add the WAY-316606 (150mg) and Misoprostol (150mg).
- The Finisher: Add the Dinoprostone (150mg) last. It is the most fragile molecule in the stack and should be handled with minimal exposure to room light.
- Filtration (Optional but Recommended): If using crushed tablets, the solution should be passed through a 0.22-micron syringe filter to remove fillers, binders, and potential contaminants.
Because this solution contains Dinoprostone and Misoprostol, it is highly susceptible to degradation from UV light and temperature fluctuations.
- The Container: You must use an Amber or Cobalt Blue Glass Bottle. Prostaglandins are photosensitive; clear glass will lead to a loss of potency within days.
- The Temperature: Store the bottle in a cool, dark place (ideally 15°C–25°C). While some prostaglandins are refrigerated, DMSO freezes at 18.5°C (65.3°F). If it freezes, you must thaw it slowly at room temperature; do not use a microwave or high heat, as this will destroy the BMP-2 (Fluvastatin) and PGE2 signals.
- Shelf Life: In research settings, a DMSO-based solution with these components typically maintains peak potency for 30–60 days. After this, the prostaglandins may begin to degrade into inactive metabolites.
3) All pharmaceutical options
I. The Wnt-Pathway & Sclerostin Axis
These compounds target the master signaling pathway for bone volume.
- Lithium Orotate: GSK-3β inhibitor; prevents the degradation of β-catenin to lock the Wnt signal to being on.
- Roflumilast (Daliresp): High-potency PDE4 inhibitor; increases cAMP to suppress the SOST (Sclerostin) gene.
- Apremilast (Otezla): Secondary PDE4 inhibitor; reduces the Sclerostin.
- Pentoxifylline (PTX): Non-selective PDE inhibitor; amplifies mechanical loading signals and suppresses Sclerostin.
- WAY-316606: Selective sFRP-1 inhibitor; neutralizes Wnt-sponges to allow unimpeded receptor binding.
- Romosozumab (Evenity): Monoclonal antibody; the strongest known direct Sclerostin inhibitor (Injectable).
- Vanadyl Sulfate: Wnt-mimetic; acts as a phosphatase inhibitor to stabilize bone-building signals.
II. The Prostaglandin (PGE) Axis
These drive Periosteal Apposition (outward expansion of the bone frame).
- Misoprostol (Cytotec): PGE1 analogue; cross-reacts with EP4 receptors for forced widening.
- Dinoprostone (PGE2): High-affinity biological signal for EP4/EP2-mediated widening.
- 16,16-Dimethyl PGE2: Stabilized, long-acting PGE2 analogue used in high-potency research.
- Alprostadil (PGE1): Directly linked to cortical hyperostosis (bone thickening).
- ONO-4819 (Rivenprost): Highly selective EP4 agonist for targeted jaw widening.
- CP-533,536: Selective EP2 agonist; triggers the initial modeling response on bone surfaces.
III. The BMP-2 & Osteoinductive Axis
These stimulate Osteoinduction (creating bone where none exists).
- Fluvastatin: High-potency statin for inducing Bone Morphogenetic Protein-2 (BMP-2).
- Simvastatin: The Gold Standard research statin for local BMP-2 induction.
- Lovastatin: The original osteogenic statin identified for skeletal signaling.
- Fenofibrate: PPAR-alpha agonist; upregulates BMP-2 and BMP-4 for cortical thickness.
- Teriparatide (Forteo): Recombinant PTH; the only purely anabolic FDA-approved bone-builder (Injectable).
IV. The Androgen & Masculinization Axis
These drive the sexual dimorphism of the facial structure.
- Testosterone Enanthate: The primary substrate for male skeletal development.
- Trenbolone: High AR-affinity steroid; drives massive localized IGF-1 and mineral density.
- Oxandrolone (Anavar): Supports collagen synthesis and dry bone mineral content.
- Mesterolone (Proviron): Lowers SHBG to increase free androgens for bone binding.
- L-Carnitine L-Tartrate (LCLT): Upregulates Androgen Receptor (AR) density.
- Boron: Lowers SHBG and supports free testosterone levels.
- Raloxifene (Evista): SERM; bone-estrogen agonist for cortical thickness and Free T boost.
- Icaritin / Epicatechin: AR stabilizers and Myostatin inhibitors.
V. The Mechanotransduction Axis
These make the bone "hyper-sensitive" to mechanical stimulus (Massage Gun).
- Amiloride: ASIC/ENaC channel blocker; sensitizes the Piezo1 pressure sensors.
- Benzamil: Potent amiloride analogue for high-level mechanosensing research.
- Donepezil (Aricept): AChE inhibitor; downregulates RANKL to prevent bone loss post-stimulus.
- Dipyridamole: Adenosine reuptake inhibitor; elevates A2AR signals for surface area expansion.
VI. The GH/IGF-1 & Sleep Axis
These maximize the nocturnal window when the bone actually builds.
- Clonidine: Alpha-2 agonist; stimulates the pituitary for endogenous GH pulses.
- Gabapentin: Increases Slow-Wave Sleep (SWS) duration for GH release.
- Baclofen: Enhances deep sleep quality and neuro-osteogenic recovery.
- LDN (Low-Dose Naltrexone): Triggers a Growth Factor Rebound during sleep.
- Telmisartan: PPAR-gamma agonist; protects IGF-1 and improves insulin sensitivity.
- Guanfacine: Selective Alpha-2A agonist for prefrontal and growth support.
VII. The Matrix & Foundation Axis
- Vitamin MK-4 (Menatetrenone): Pharmaceutical-grade collagen cross-linker.
- Strontium Citrate: Replaces calcium in the lattice for increased lattice volume.
- Piascledine (ASU): Upregulates TGF-β for a thicker collagen scaffold.
- Ipriflavone: Synthetic isoflavone for direct Type I Collagen synthesis.
- Methylene Blue (USP): Mitochondrial respirator; provides ATP fuel for remodeling.
- Hydrochlorothiazide (HCTZ): Promotes renal calcium retention for mineralization.
- Nebivolol / Tadalafil: Increases Nitric Oxide (NO) for bone angiogenesis.
VIII. Potential Future Candidates (Niche Research)
- Fasudil: Rho-kinase (ROCK) inhibitor; forces stem cells to become bone instead of fat.
- Mequinol: Potential local carrier/modulator (clarified as distinct from bone-builders).
- Catalpol / Geniposide: Iridoid glycosides for neuro-osteogenic jaw coupling.
- Nafamostat Mesilate: Potent ASIC blocker for mechanical sensitization.
- Celecoxib (Selective COX-2): (Used cautiously in research as a comparator, as it typically inhibits growth).
How to use Steroids
For this example we are going to be using testosterone enanthate 500mg cycle for 20 weeks. This guide includes How to start using androgens, what to do while using androgens and how to safely come off clean when wanted. 20-week cycle of 500 mg Testosterone Enanthate, the focus is on maintaining stable blood levels, managing estrogen effectively to support bone growth, and ensuring a safe transition at both the start and end of the cycle.
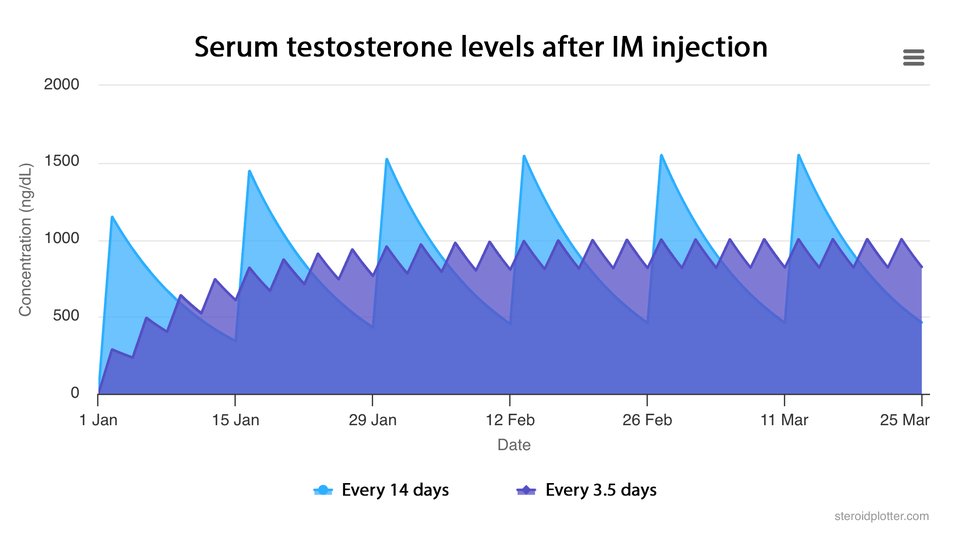
1. Dosing Schedule: The 3.5-Day Split
To minimize the "peaks and troughs" associated with large weekly injections, splitting the 500 mg dose into two 250 mg injections is a common research-backed strategy.
- Optimal Days: The most consistent schedule is every 3.5 days.
- Example Routine: Inject Monday morning and Thursday evening. This ensures your blood levels remain stable, which can reduce side effects like acne and mood swings.
2. Aromatase Inhibitor (AI): Exemestane (Aromasin)
To meet your goal of preventing gynecomastia and reducing adipose estrogen while protecting bone health, Exemestane (Aromasin) is often preferred over Anastrozole (Arimidex).
- Bone Health: Estrogen is vital for male bone mineral density. Unlike non-steroidal inhibitors, Aromasin is a "suicidal" inhibitor that may be less detrimental to bone metabolism when dosed correctly.
- Selective Suppression: It effectively prevents the aromatization of testosterone into estrogen in peripheral tissues like adipose (fat).
- Androgenic Metabolite: Exemestane breaks down into 17-hydroexemestane, which has a weak affinity for the androgen receptor and has been shown in studies to be bone-protective.
- IGF-1 Preservation: Unlike other AIs, Exemestane is less likely to negatively impact your IGF-1 levels at moderate doses.
- Dosing: A common starting point is 12.5 mg (half a tablet) twice weekly, taken on the same days as your injections. Adjust based on blood work to ensure estrogen isn't crushed, as very low estrogen can lead to joint pain and brittle bones.
| E2 Target | 20–30 pg/mL |
3. Tapering Protocol: The Beginning and End
Tapering is used to help the body adjust to exogenous hormones and, more importantly, to restart natural production after the cycle.
- The Beginning (Front-loading): Most do not taper up but rather start at the full dose. Because Enanthate is a long ester, it takes 3–4 weeks to reach peak concentrations.
- The End (Tapering Off): Abruptly stopping 500 mg of testosterone is not advisable. A typical taper involves reducing the dose by 25% every two weeksduring the final month of the cycle.
- Weeks 17–18: Reduce to 375 mg/week.
- Weeks 19–20: Reduce to 250 mg/week
- As you taper your Testosterone at the end of the 20 weeks (reducing from 500mg to 250mg), you must proportionally reduce the AI dose.
- The Goal: You want to avoid an Estrogen Crash during the transition into PCT, which would make your bones brittle and kill your morale.
4. Essential Ancillaries for a 20-Week Cycle
A 20-week cycle is considered long, making organ and hormonal support critical.
- HCG (Human Chorionic Gonadotropin): To prevent testicular atrophy and make Post-Cycle Therapy (PCT) more effective, research suggests 250–500 IU twice weekly throughout the cycle.
- Telmisartan: 40mg–80mg daily.
Goal: Blocks Angiotensin-II to prevent facial edema (moon face) and manages blood pressure.
HCTZ (Hydrochlorothiazide): 12.5mg daily.
Goal: Flushes water from the face (adipose) while promoting renal calcium retention for the bone matrix. - Myo-Inositol & D-Chiro Inositol (40:1): 2g–4g daily.
Goal: Improves insulin sensitivity for IGF-1 efficiency and provides secondary aromatase modulation. - Pentoxifylline (PTX): 400mg ER (Extended Release) twice daily.
Goal: Essential for 500mg Test users; it manages the increased hematocrit (blood thickening) and amplifies mechanical bone signals. - Nebivolol: 5mg daily.
Goal: Heart rate stability and Nitric Oxide (NO) release for bone angiogenesis. - Tadalafil: 5mg daily.
Goal: Chronic PDE5 inhibition for vasodilation and localized bone blood flow. - minoxidil: Prevent hair loss from increased DHT
- Isotretinoin: Acne, oil production
Long-term usage of exogenous hormones necessitates a structured cessation.
- Wait Period: Allowing time for the long-acting Testosterone esters to clear the system.
- The Restart (Wks 23-28): Involves the use of Selective Estrogen Receptor Modulators like Nolvadex to help restore natural testosterone production.
- Post-Cycle Therapy (PCT): Wait 14–18 days after your last injection before starting PCT. A standard protocol includes: Tamoxifen (Nolvadex): 20 mg daily for 4–6 weeks. Clomiphene (Clomid): 25–50 mg daily for 4 weeks.
this is a good video tutorial on how to inject test. as for the needles, it depends on which testosterone and dose ur using. REMEMBER USE AN MCT OIL CARRIER BASE
Thanks everyone for Reading. this post does not have a TL;DR. If you guys have any questions please feel free to ask. I request you not to spread hate.
Attachments
-
 1774164813557.png38.8 KB · Views: 0
1774164813557.png38.8 KB · Views: 0 -
 1774180744918.gif43 bytes · Views: 0
1774180744918.gif43 bytes · Views: 0 -
 1774180757697.gif43 bytes · Views: 0
1774180757697.gif43 bytes · Views: 0 -
 1774790137296.png1.1 KB · Views: 0
1774790137296.png1.1 KB · Views: 0 -
 1774790451832.png1.4 KB · Views: 0
1774790451832.png1.4 KB · Views: 0 -
 1774790491584.png2.9 KB · Views: 0
1774790491584.png2.9 KB · Views: 0 -
 1774790717827.png1.3 KB · Views: 0
1774790717827.png1.3 KB · Views: 0 -
 1774180573637.gif43 bytes · Views: 0
1774180573637.gif43 bytes · Views: 0 -
 1774180417080.gif43 bytes · Views: 0
1774180417080.gif43 bytes · Views: 0 -
 1774164869448.png61.2 KB · Views: 0
1774164869448.png61.2 KB · Views: 0 -
 1774167169351.gif43 bytes · Views: 0
1774167169351.gif43 bytes · Views: 0 -
 1774168172285.gif43 bytes · Views: 0
1774168172285.gif43 bytes · Views: 0 -
 1774175738304.gif43 bytes · Views: 0
1774175738304.gif43 bytes · Views: 0 -
 1774175812122.gif43 bytes · Views: 0
1774175812122.gif43 bytes · Views: 0 -
 1774175821221.gif43 bytes · Views: 0
1774175821221.gif43 bytes · Views: 0 -
 1774175910501.gif43 bytes · Views: 0
1774175910501.gif43 bytes · Views: 0 -
 1774175910509.gif43 bytes · Views: 0
1774175910509.gif43 bytes · Views: 0 -
 1774178842642.gif43 bytes · Views: 0
1774178842642.gif43 bytes · Views: 0 -
 1774180417087.gif43 bytes · Views: 0
1774180417087.gif43 bytes · Views: 0 -
 1774180417094.gif43 bytes · Views: 0
1774180417094.gif43 bytes · Views: 0


 The core problem with this whole theory
The core problem with this whole theory Where the write-up sounds scientific but breaks down
Where the write-up sounds scientific but breaks down Net effect: you’re more likely to damage bone than grow it
Net effect: you’re more likely to damage bone than grow it The biggest risks in this protocol
The biggest risks in this protocol What actually can influence facial structure
What actually can influence facial structure Bottom line
Bottom line